
pharma solutions
The Morris Group creates GxP applications on Smartsheet as an economical alternative to expensive commercial software. Our templates are FDA compliant and can be implemented in a fraction of the time and cost of commercial systems.
Small pharma and medical device companies often resort to Excel, SharePoint and Access to avoid paper records. Unfortunately, this approach can create more problems than it solves.
We can help you replace hundreds of spreadsheets, PDFs and other flat files with a secure database that is 21 CFR Part 11 compliant, thereby reducing regulatory risk.
Our out of the box templates include:
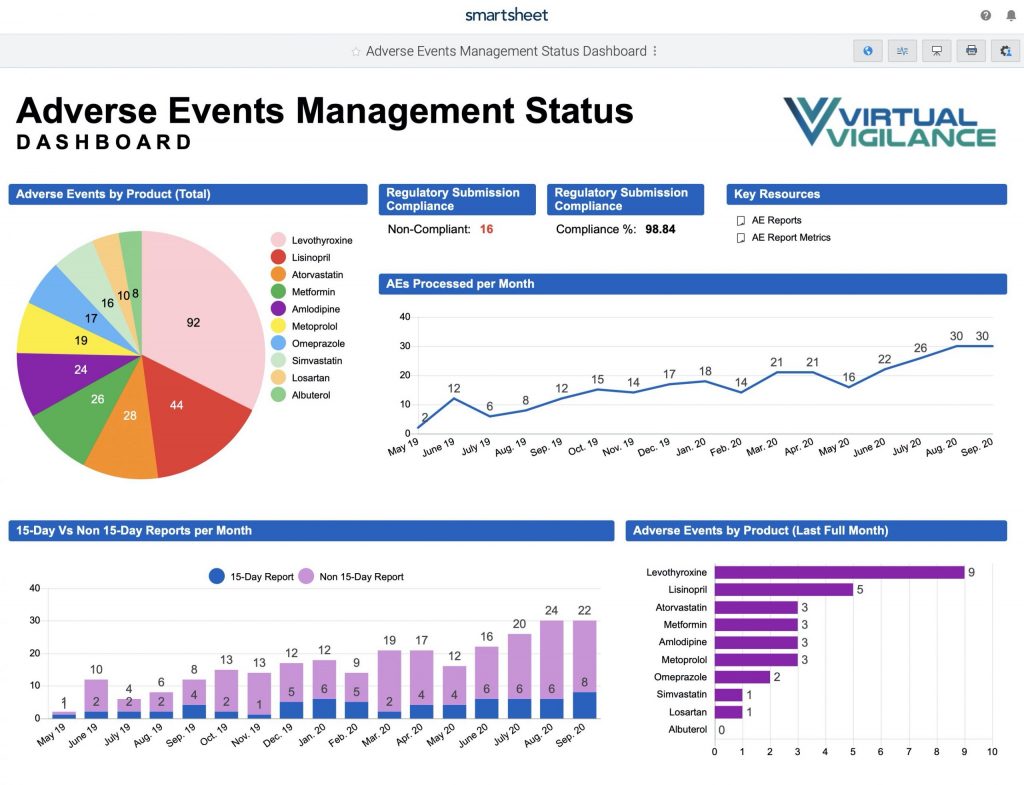
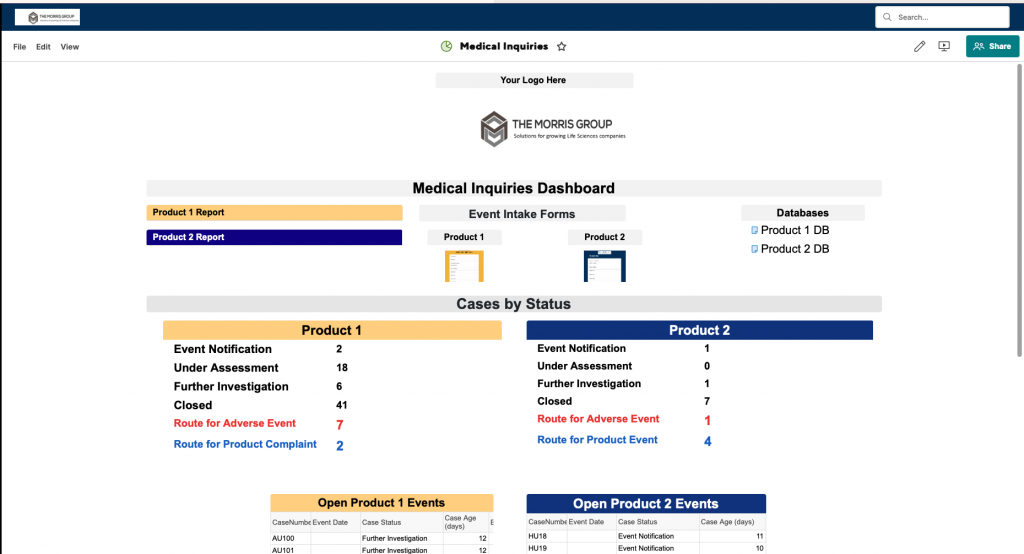
- Adverse Events
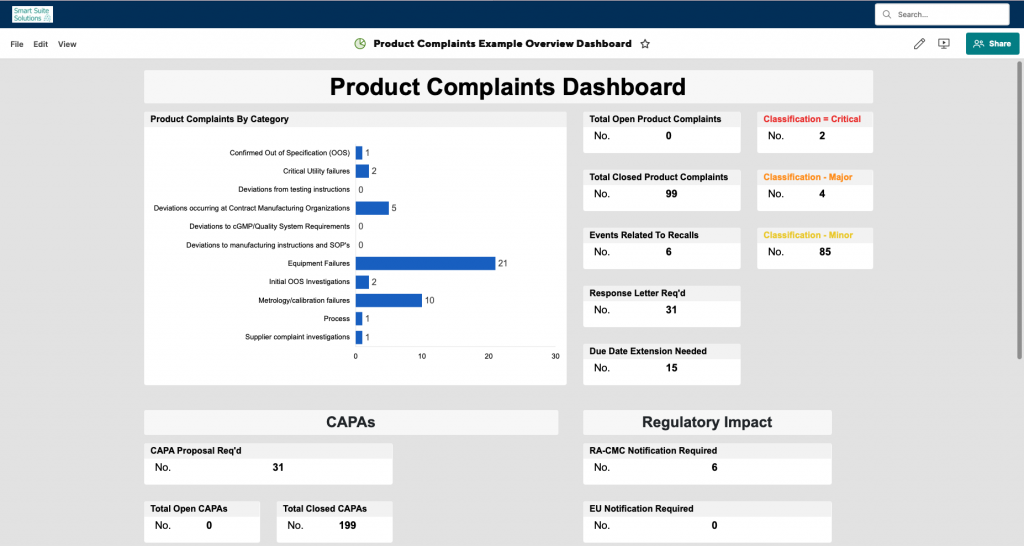
- Product Complaints
- Clinical Trial Management
- Change Control
- Quality Event Management
Get In Touch
info@themorris-group.com
Location
Fort Myers, FL
